 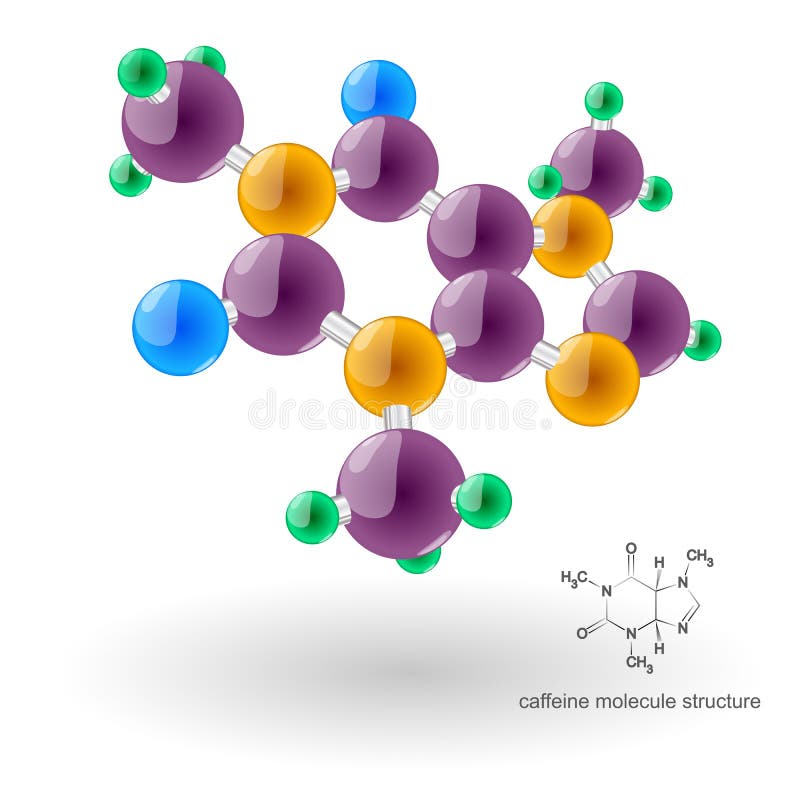
Caffeine Melting Point Standard, pharmaceutical secondary standard traceable to USP, Melting range. Contributors William Reusch, Professor Emeritus ( Michigan State U. Caffeine structure data file available for download. It has a density of 1.2g/cm3 with a volatility of 0.5 and. The last compound, an isomer of octane, is nearly spherical and has an exceptionally high melting point (only 6 below the boiling point). Caffeine tastes bitter and the density is 1.23 g mL -1 and its melting point is 235 ✬ and at higher temperatures, it decomposes. In addition, the attractive interaction between dipoles falls off much more rapidly with increasing distance than do the ion–ion interactions. Caffeine has a boiling point of 178oC and a melting point of 238oC. The molecule is the smallest observable group of uniquely bonded atoms that represent the composition, configuration and characteristics of a pure compound. There are 24 atoms and 4 elements in caffeine, therefore containing 8. \): Both Attractive and Repulsive Dipole–Dipole Interactions Occur in a Liquid Sample with Many Moleculesīecause each end of a dipole possesses only a fraction of the charge of an electron, dipole–dipole interactions are substantially weaker than the interactions between two ions, each of which has a charge of at least ☑, or between a dipole and an ion, in which one of the species has at least a full positive or negative charge. Get Caffeine molecular formula, CAS number, boiling point, melting point, applications, synonyms and more here. Caffeine has no odor, a density of 1.23 g/cm3 and a boiling point of 352 (177.8 ). 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
